Organic Electronics & Materials Research Laboratory
Department of Physics & Astrophysics, University of Delhi, India
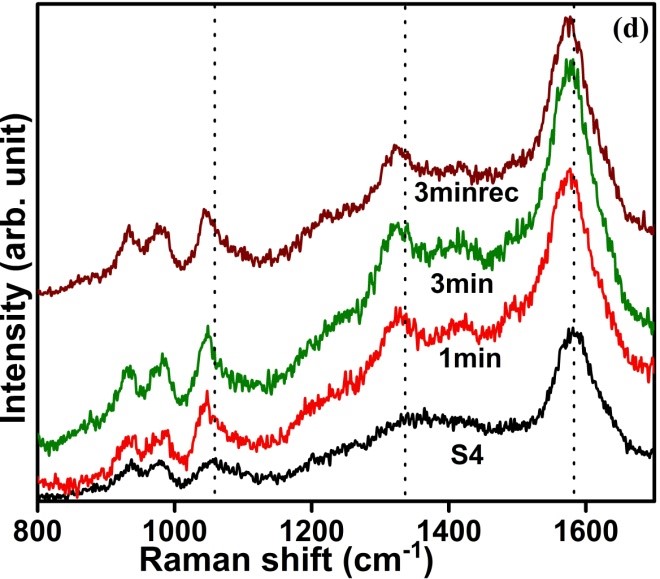
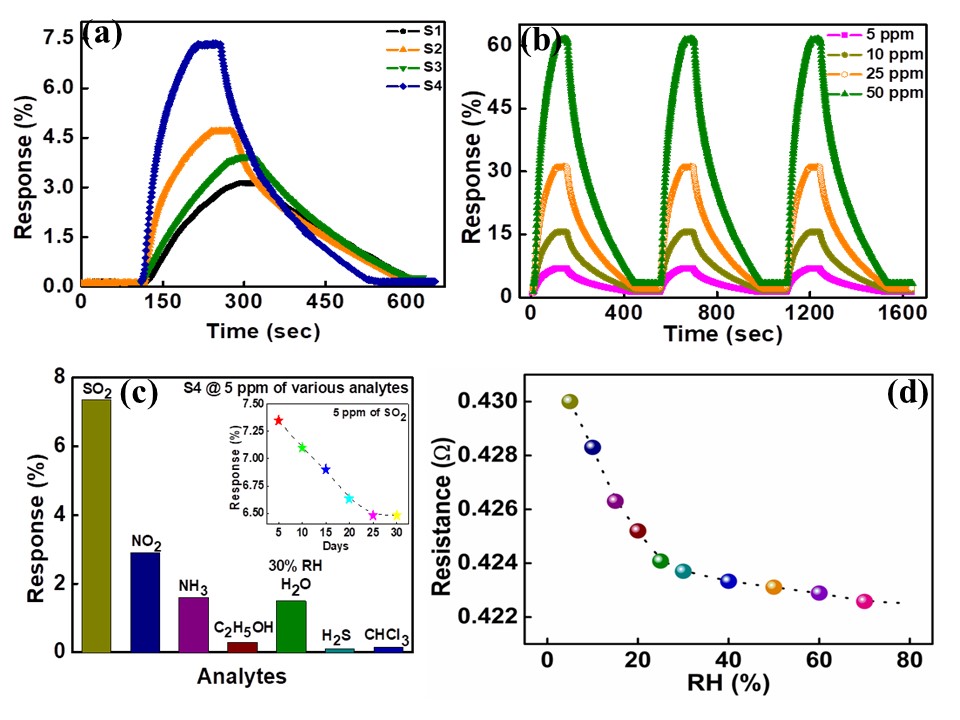
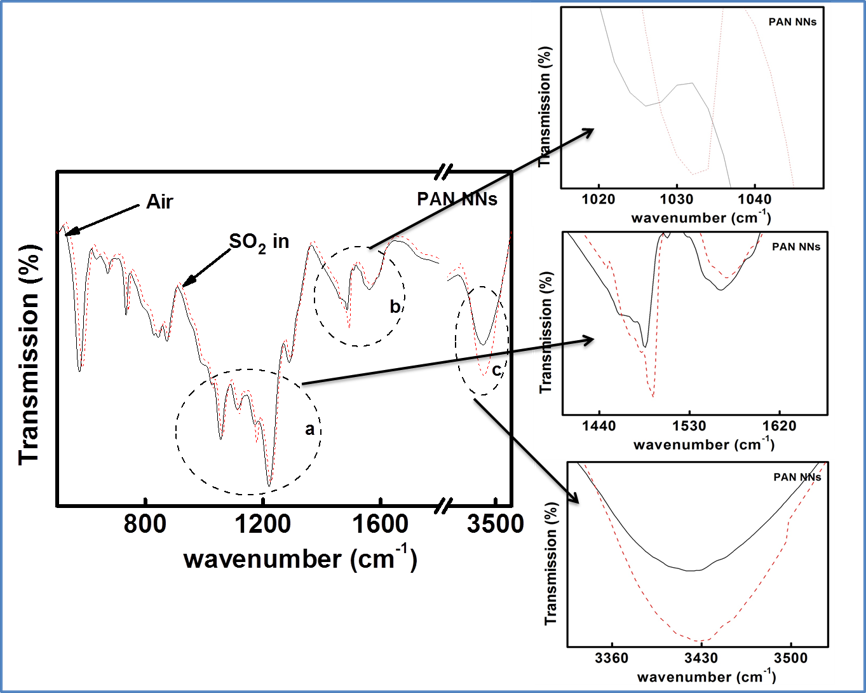
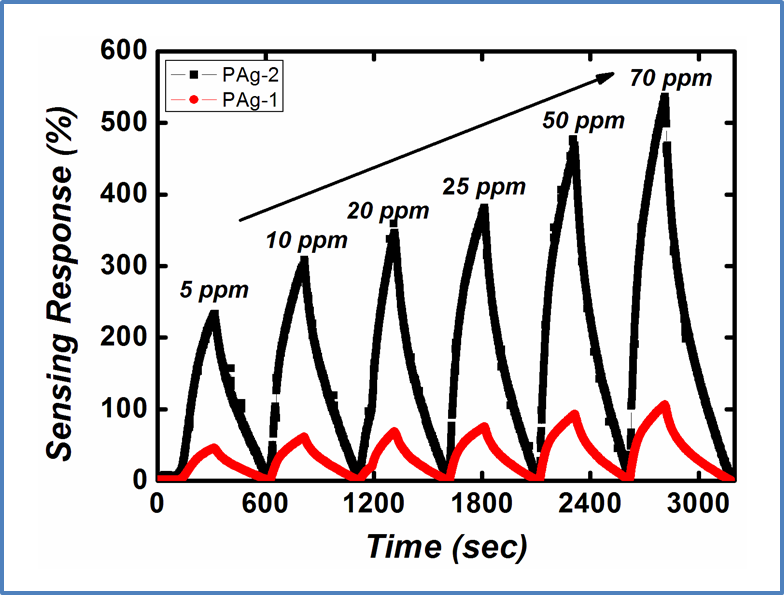
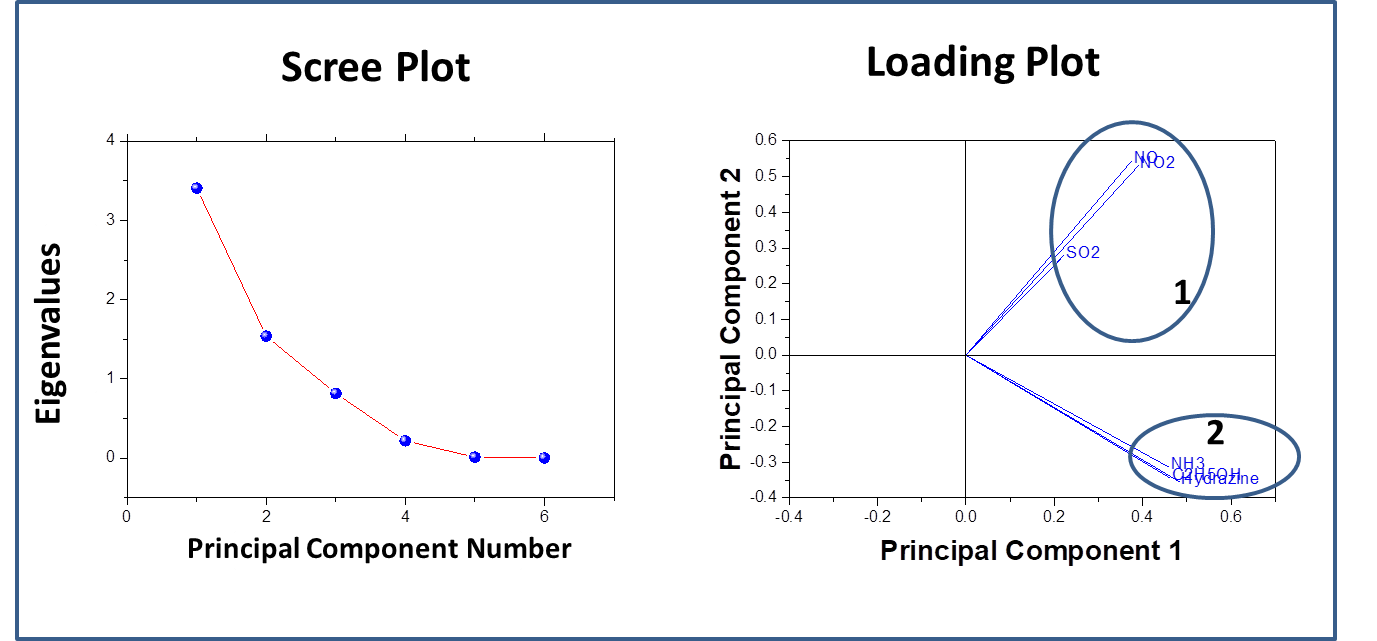
Recent years have witnessed an increasing demand towards the development of simple and effective gas sensors, to be progressively employed in mass-market applications such as indoor air quality control, environmental monitoring, medical diagnosis, and industrial safety as well as in the areas of explosive and toxic gas detection.The conventional gas detection techniques such as gas chromatography, infrared monitoring, mass spectrometry and acoustic methods are known to give accurate measurements and good performance. However, the devices based on these technologies usually have bulky configuration, high fabrication cost and consume a lot of power. As an alternative, nanomaterial-based solid state gas sensing devices are attaining prevalence due to their ease of operation, compact configuration and low fabrication cost. In our work on gas sensing materials, we employ the chemiresistive gas sensing mechanism. Most of the conventional gas sensors are based on metal-oxide semiconductors such as ZnO, SnO2, NiO, etc, which are low-cost, exhibit good sensitivity and fast response/recovery. However, they require to be operated at higher temperatures (>200 ᵒC). Therefore, require heating assembly for their operation. In order to do away with the limitation of high operating temperature and achieve gas sensing response at room temperature, our group has been working towards the development of gas sensing devices based on conducting polymers like polyaniline, polypyrrole, polyanisidine and their nanocomposites with metal oxides such as polyaniline-tungsten oxide. In one of the novel works by the group, different morphologies of polyaniline were synthesized using surfactant-assisted approach, which showed excellent room temperature response towards low ppm concentration of SO2 gas. Nanostructures of polypyrrole-tin oxide have also been investigated for gas sensing response towards ammonia, using Kelvin-probe force microscopy. Although, polymers exhibit a promising performance as room temperature gas sensing materials, there is often a consideration of their long-term stability. In view of this, more recently, our group has explored reduced graphene oxide (rGO) as the active layer for gas sensing at room temperature due to its extraordinary electronic properties as well as good thermal and ambient stability. Graphene oxide prepared in our lab, is reduced through chemical or thermal route, followed by ion-beam irradiation. We are also working towards synthesis of sensing materials based upon nanocomposites of reduced graphene oxide and metal oxide semiconductors with the motivation that such hybrid nanostructures tend to give better response due to synergistic effects at the heterojunction sites. Nanocomposites of rGO and ZnO have been synthesized and examined to give fast and selective response towards ammonia at room temperature. Nevertheless, these materials have high specific surface area, but their structure could still be made more porous to improve the sensitivity of devices. A new opportunity to address this problem has recently emerged with the possibility of combination of rGO and metal organic frameworks (MOFs). While studying the response of the materials towards various gases in our lab, the materials are tested for their sensitivity, selectivity, stability in ambient conditions. Prototype devices are prepared for monitoring day to day performance of these sensors based on the prepared materials.
Published and ongoing work
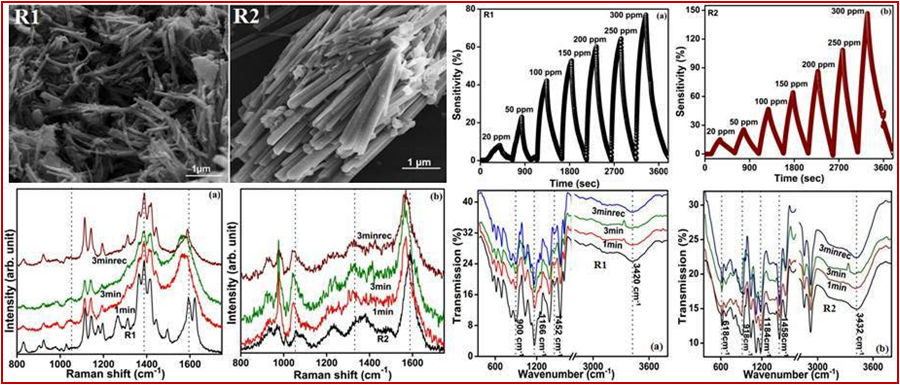

Polypyrrole Nanotubes
Approach used : Dye assisted chemical oxidative polymerisation at room temperature
Approach used : Dye assisted chemical oxidative polymerisation at room temperature

Polypyrrole Nanowires
Approach used : Dye assisted chemical oxidative polymerisation at room temperature (varying dye cmc)
Approach used : Dye assisted chemical oxidative polymerisation at room temperature (varying dye cmc)

Polypyrrole Nanorods
Approach used : Anionic Surfactant assisted chemical oxidative polymerization at room temperature
Approach used : Anionic Surfactant assisted chemical oxidative polymerization at room temperature

Polypyrrole Nanoparticles
Approach used : CTAB assisted chemical oxidative polymerization
Approach used : CTAB assisted chemical oxidative polymerization

Polypyrrole No-nanostructure
Approach used : HClO4 assisted chemical oxidative polymerisation
Approach used : HClO4 assisted chemical oxidative polymerisation
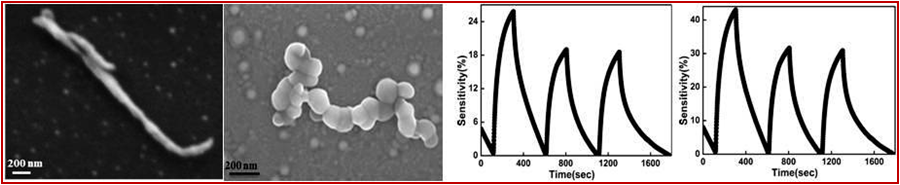
Polypyrrole Nanofibers
Approach used : SDS assisted chemical oxidative polymerisation
Approach used : SDS assisted chemical oxidative polymerisation

Polypyrrole Nanoparticles
Approach used : H2SO4 assisted chemical oxidative polymerisation
Approach used : H2SO4 assisted chemical oxidative polymerisation

Polypyrrole Nanorods
Approach used : SDS assisted chemical oxidative polymerisation
Approach used : SDS assisted chemical oxidative polymerisation

Polypyrrole Nanoparticles
Approach used : SDS assisted chemical oxidative polymerisation
Approach used : SDS assisted chemical oxidative polymerisation

Polypyrrole Nanofibers
Approach used : SDS assisted chemical oxidative polymerisation
Approach used : SDS assisted chemical oxidative polymerisation

Polypyrrole Nanofibers
Approach used : Oxidant assisted chemical oxidative polymerisation
Approach used : Oxidant assisted chemical oxidative polymerisation

Polypyrrole No-nanostructure
Approach used : HNO3assisted chemical oxidative polymerisation
Approach used : HNO3assisted chemical oxidative polymerisation

Polypyrrole No-nanostructure
Approach used : HClO4 assisted chemical oxidative polymerisation
Approach used : HClO4 assisted chemical oxidative polymerisation

Polypyrrole Nanoparticles
Approach used : SDS assisted chemical oxidative polymerisation
Approach used : SDS assisted chemical oxidative polymerisation

Polypyrrole Nanofibers
Approach used : H2SO4 assisted chemical oxidative polymerisation
Approach used : H2SO4 assisted chemical oxidative polymerisation

Polypyrrole Nanoparticles
Approach used : SDS assisted chemical oxidative polymerisation
Approach used : SDS assisted chemical oxidative polymerisation

Polypyrrole Nanoparticles
Approach used : SDS assisted chemical oxidative polymerisation
Approach used : SDS assisted chemical oxidative polymerisation

Bulk Polyaniline
Approach used : Conventional synthesis: Simple chemical oxidative polymerisation at room temperature
Approach used : Conventional synthesis: Simple chemical oxidative polymerisation at room temperature
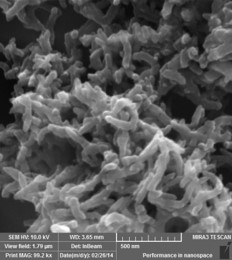
Polyaniline
Approach used : V2O5 assisted chemical oxidative polymerisation
Approach used : V2O5 assisted chemical oxidative polymerisation

Polyaniline
Approach used : AgNO3 assisted chemical oxidative polymerisation
Approach used : AgNO3 assisted chemical oxidative polymerisation

Polyaniline
Approach used : Organic Acid assisted chemical oxidative polymerisation
Approach used : Organic Acid assisted chemical oxidative polymerisation
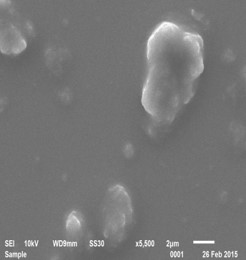
Polyaniline
Approach used : Organic Acid assisted chemical oxidative polymerisation
Approach used : Organic Acid assisted chemical oxidative polymerisation

Polyaniline
Approach used : Organic Acid assisted chemical oxidative polymerisation
Approach used : Organic Acid assisted chemical oxidative polymerisation

Polyaniline Nanoparticles
Approach used : Organic Acid assisted chemical oxidative polymerisation
Approach used : Organic Acid assisted chemical oxidative polymerisation
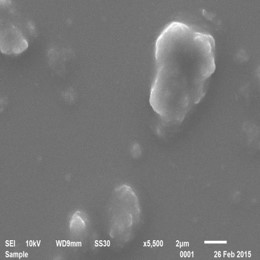
Bulk Polyaniline
Approach used : Organic Acid assisted chemical oxidative polymerisation
Approach used : Organic Acid assisted chemical oxidative polymerisation

Polyaniline Nanospheres
Approach used : Organic Acid assisted chemical oxidative polymerisation
Approach used : Organic Acid assisted chemical oxidative polymerisation

Polyaniline Nanospheres
Approach used : CTAB assisted chemical oxidative polymerisation (variation in CTAB concentration)
Approach used : CTAB assisted chemical oxidative polymerisation (variation in CTAB concentration)

Polyaniline Nanofibres
Approach used : Template less low temperature (-5ᵒC) Chemical oxidative polymerisation
Approach used : Template less low temperature (-5ᵒC) Chemical oxidative polymerisation

Polyaniline Nanospheres
Approach used : CTAB assisted chemical oxidative polymerisation (variation in CTAB concentration)
Approach used : CTAB assisted chemical oxidative polymerisation (variation in CTAB concentration)
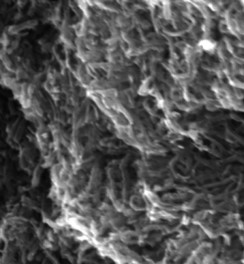
Polyaniline
Approach used : CTAB assisted chemical oxidative polymerisation (variation in CTAB concentration)
Approach used : CTAB assisted chemical oxidative polymerisation (variation in CTAB concentration)

Polyaniline Nanoneedles
Approach used : CTAB assisted chemical oxidative polymerisation (variation in CTAB concentration)
Approach used : CTAB assisted chemical oxidative polymerisation (variation in CTAB concentration)

Polyaniline
Approach used : Non-ionic Surfactant assisted chemical oxidative polymerisation
Approach used : Non-ionic Surfactant assisted chemical oxidative polymerisation
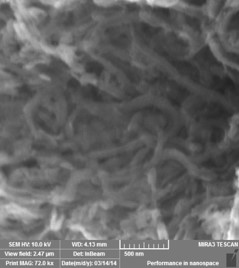
Polyaniline Nanofibres
Approach used : Cationic Surfactant assisted chemical oxidative polymerisation
Approach used : Cationic Surfactant assisted chemical oxidative polymerisation

Polyaniline Nanoparticles
Approach used : FeCl3assisted chemical oxidative polymerisation
Approach used : FeCl3assisted chemical oxidative polymerisation
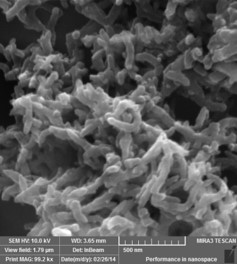
Polyaniline Nanofibres
Approach used : V2o5 assisted chemical oxidative polymerisation
Approach used : V2o5 assisted chemical oxidative polymerisation

Polyaniline Nanoflakes
Approach used : AgNO3 assisted chemical oxidative polymerisation
Approach used : AgNO3 assisted chemical oxidative polymerisation

Polypyrrole Nanoparticles Nanoparticles
;
Approach used : Organic Acid assisted chemical oxidative polymerisation
Approach used : Organic Acid assisted chemical oxidative polymerisation
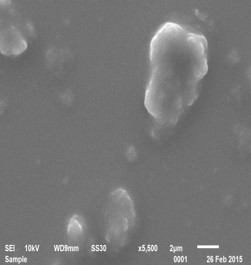
Bulk Polyaniline
Approach used : Organic Acid assisted chemical oxidative polymerisation
Approach used : Organic Acid assisted chemical oxidative polymerisation

Polyaniline Nanospheres
Approach used : Organic Acid assisted chemical oxidative polymerisation
Approach used : Organic Acid assisted chemical oxidative polymerisation

Polyaniline Nanofibres
Approach used : Organic Acid assisted chemical oxidative polymerisation
Approach used : Organic Acid assisted chemical oxidative polymerisation

Polyaniline Nanorods
Approach used : Inorganic Acid assisted chemical oxidative polymerisation
Approach used : Inorganic Acid assisted chemical oxidative polymerisation
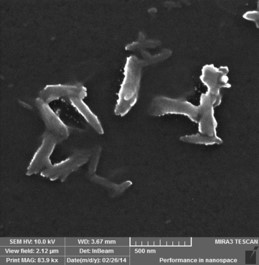
Polyaniline Nanotubes
Approach used : Inorganic Acid assisted chemical oxidative polymerisation
Approach used : Inorganic Acid assisted chemical oxidative polymerisation

Polyaniline Nanoparticles
Approach used : Inorganic Acid assisted chemical oxidative polymerisation
Approach used : Inorganic Acid assisted chemical oxidative polymerisation
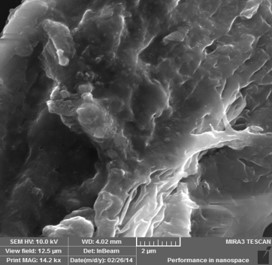
Bulk Polyaniline
Approach used : Oleic acid assisted chemical oxidative polymerisation
Approach used : Oleic acid assisted chemical oxidative polymerisation